Dec. 3 WEBINAR | Leveraging Embase AI in life science research with Dr. Vassiki Sanogo and Regina Maxwell, MLIS

Unfortunately we don't fully support your browser. If you have the option to, please upgrade to a newer version or use Mozilla Firefox, Microsoft Edge, Google Chrome, or Safari 14 or newer. If you are unable to, and need support, please send us your feedback.
We'd appreciate your feedback.Tell us what you think!
Are you missing critical information for medical device compliance and development? Embase provides targeted medical device literature searches through broad content coverage, precise indexing and device-specific terms.


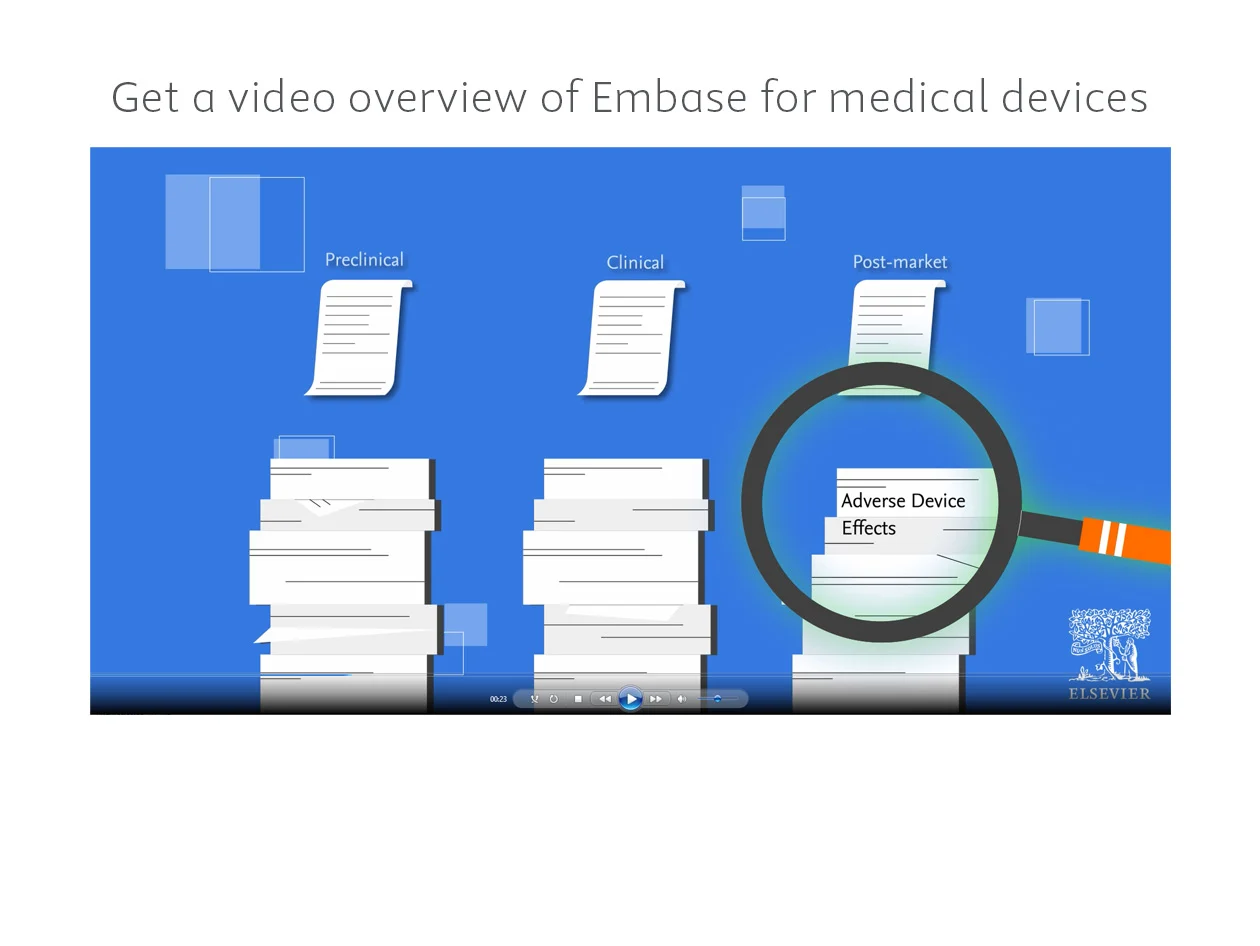
Embase has the medical device research, evidence and literature monitoring tools to help you:
Develop and validate new concepts
Comply with medical device regulations
Prepare clinical evaluation reports for CE Marketing approval
Conduct post-market surveillance

Embase takes the pain out of creating the complex search strings needed for high-recall, high-precision searches. A medical device search form and preset terms take the guesswork out of search.

Our team manually indexes full-text articles, so you can search by device, concept, manufacturer, procedure, device trade name, FDA-approved Global Medical Device Nomenclature (GMDN) terms and more.
Email alerts ensure you receive the latest in published information, including preprints and conference proceedings.

The lack of a SOTA search is one of the top reasons for MDR/IVDR non-conformance findings.
Use Embase’s search command to keep up to date with relevant SOTA content in accordance with regulatory guidance.
For MDR compliance, databases must show sufficient European reach.
Embase covers all the content contained in MEDLINE and has unique coverage for Europe, including conference abstracts and European journals.
One of the most amazing things about Embase is that the entire article is indexed for terms and product names. For medical devices, being able to locate information on a specific product is a huge plus.
Dawn McMillen
Library Resources Supervisor at Stryker Orthopedics
