
The innovator’s guide to carbon capture, utilization and storage (CCUS)
The current state and future outlook of CCUS in industry.

What is CCUS?
According to the United Nations’ Intergovernmental Panel on Climate Change (IPCC), achieving net-zero targets is no longer possible without removing a significant amount of carbon dioxideopens in new tab/window from the environment. Carbon capture, utilization and storage (CCUS) technologies remove existing carbon dioxide emissions, store them safely and effectively and provide opportunities to utilize them for future projects. These innovative technologies play a significant role in achieving the energy industry’s emission reduction goals and turning carbon waste into high-value products.
CCUS technologies are used to isolate CO2 from heavy industry emissions, inject it into permanent storage sites and utilize it in the creation of high-value products like building materials and fuel.
Carbon capture includes methods such as pre-combustion, post-combustion and oxy-combustion to sequester CO2 from industrial emissions
Carbon utilization refers to transforming captured carbon into essential products, primarily through catalysis
Carbon storage involves injecting captured CO2 into geological formations, like saline formations and depleted natural gas reservoirs, for permanent storage
With a global call for climate change initiatives, industries must invest in emission-reduction technologies to achieve net-zero emissions and lead the way in carbon removal from the environment. A host of government incentives, such as carbon credits and project grants, are already in motion to help organizations explore CCUS projects as a solution to achieve climate goals.
CCS vs. CCUS: A new focus on utilization
While carbon sequestration technology dates back to the 1920s, the idea of capturing and storing CO2 really began in the late 1970s. Since then, practical applications of the technology have focused on carbon capture and storage (CCS), while carbon utilization research carried on in the background. Both CCS and CCUS apply the same technologies to capture and store carbon, but the difference lies in what happens to the captured carbon.
Although the CCS process is designed to store captured carbon indefinitely, it is meant as a short-term solution to the problem of climate change. In contrast, CCUS projects focus on the long-term goal of utilizing captured carbon to produce a wide range of products, from hydrocarbons to cement.
There are three primary reasons for the industry’s shift toward CCUS.
Space considerations for carbon storage
CCS was meant as a stopgap measure, lasting anywhere from five to 10 years. Research shows that the earth currently has “between 8,000 and 55,000 gigatonnes (Gt) of practically accessible geologic storage capacity for carbon dioxide” (source: "Developing a Consistent Database for Regional Geologic CO2 Storage Capacity Worldwideopens in new tab/window, Energy Procedia.) But the eligibility of storage space is also affected by other factors like the suitability of the geography and the proximity to residential areas along with laws and regulations.
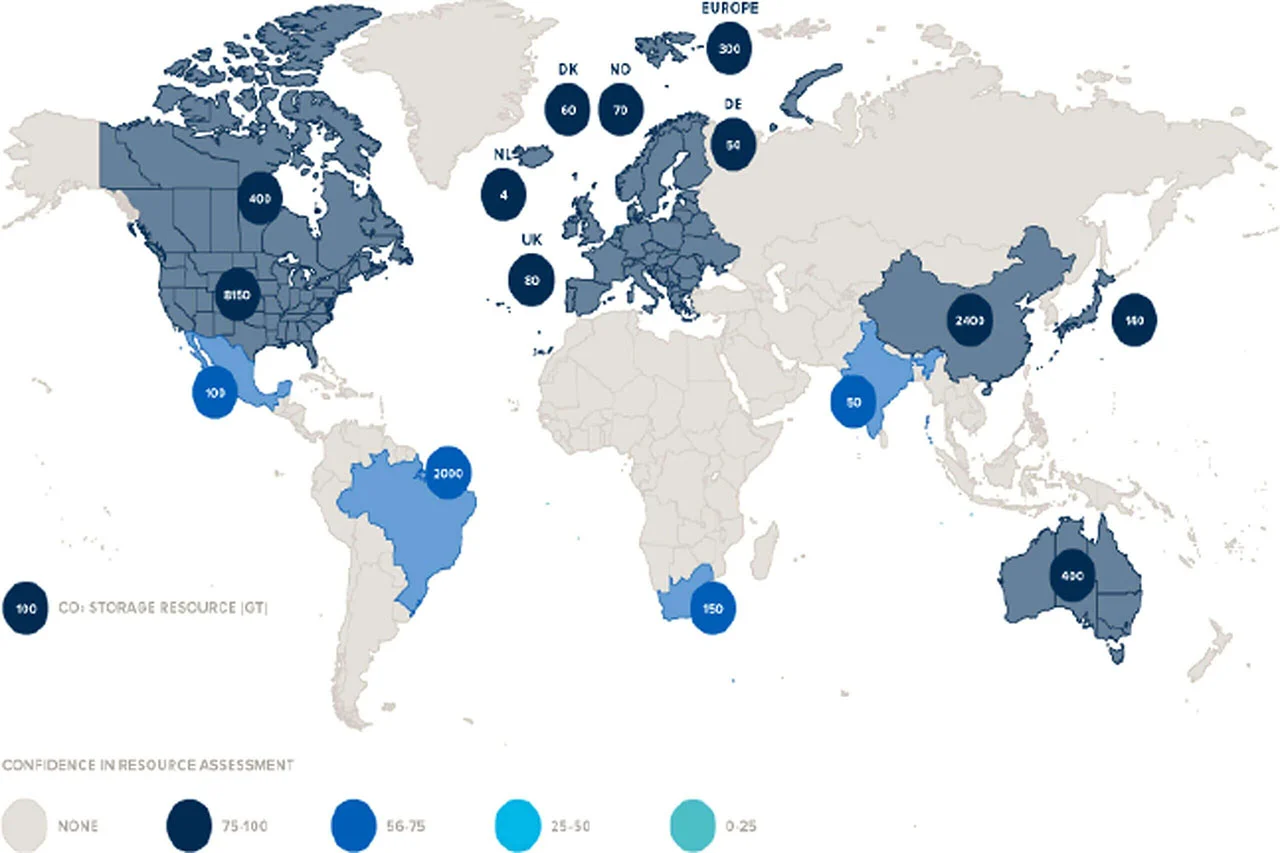
Source: "A review of large-scale CO2 shipping and marine emissions management for carbon capture, utilisation and storage" (Applied Energy, Volume 287opens in new tab/window).
Limited resources
Instead of letting captured carbon go to waste, carbon utilization has the potential to regenerate chemical feedstocks essential to the energy industry. In the future, captured carbon could be an input source for valuable products like methane, acetic acid and fuel. Although this is a longer-term issue, focusing on regenerating resources now not only reduces your environmental impact from extraction but also establishes a sustainable business model.
Economic considerations
While CCS projects support net-zero initiatives, they also require a substantial monetary commitment without much financial benefit for energy companies. The cost of building new facilities or retrofitting existing spaces may be covered initially by government grants and credits, but it’s not a sustainable solution. Utilization is a long-term financial investment because it creates a circular economyopens in new tab/window where resources are used and reused for production.
Methods of carbon capture
The three primary methods of carbon capture include post-, pre- and oxy-combustion, with post-combustion being the current industry standard.
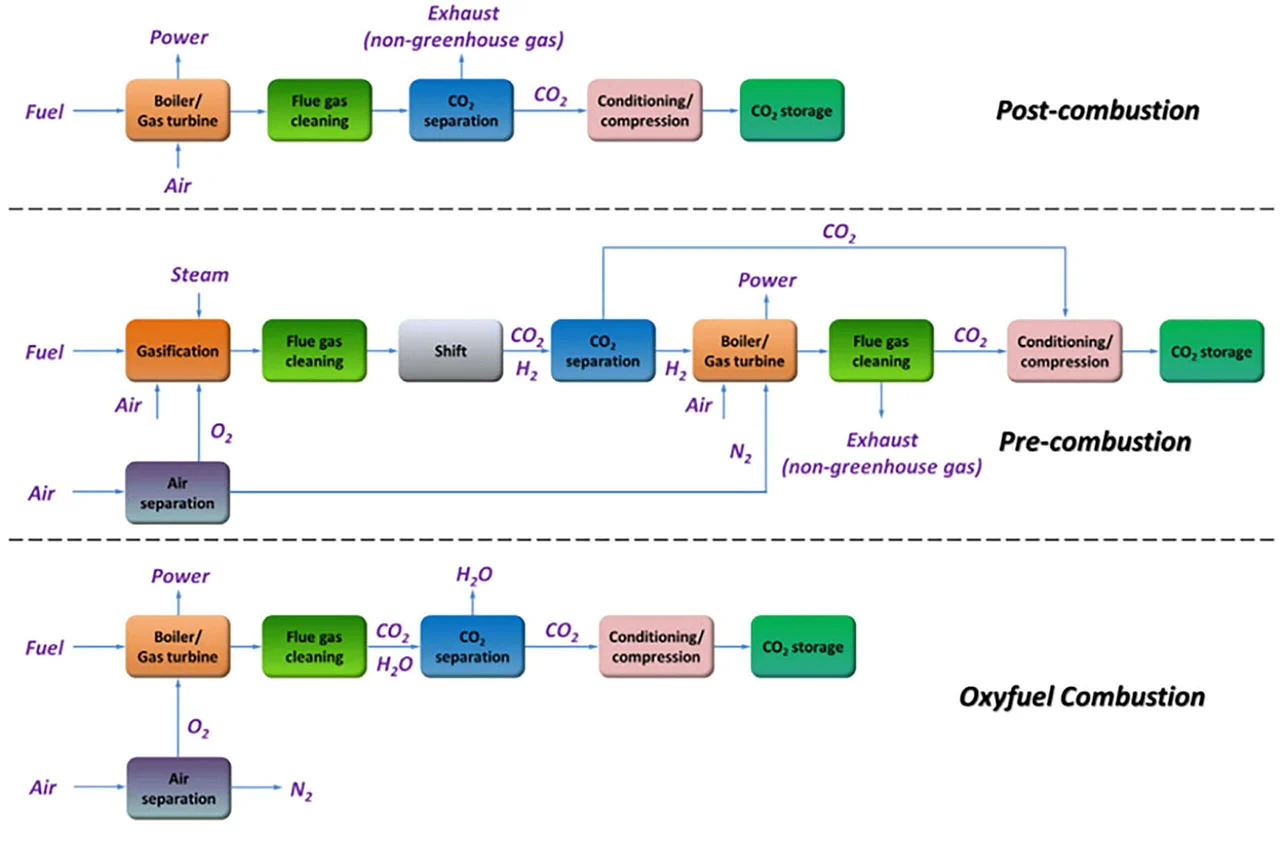
Source: "A comprehensive overview of carbon dioxide capture: From materials, methods to industrial status" (Materials Today, Volume 60opens in new tab/window).
Post-combustion capture
In post-combustion capture, CO2 is separated after combustion, most commonly with a liquid amine scrubber (known as amine gas treatment). This method uses aqueous solutions of alkylamines, or amines, to sequester CO2 from gasses produced during the refining process.
Discover more about alkylamines on ScienceDirect.opens in new tab/window
Post-combustion is the most common form of carbon capture for energy companies because it is easier to implement than pre- or oxy-combustion. Most facilities already have the required components for the process, with only small retrofits or changes required, making post-combustion the less expensive option. Post-combustion has also been thoroughly researched and practically applied for decades in the energy industry, with natural gas facilities using liquid amine scrubbers to ensure the end product is high quality.
Pre-combustion
Pre-combustion capture occurs between the pre-treatment and combustion stages, which reduces the amount of CO2 input during combustion. This process isn’t an industry standard at the moment due to cost, temperature requirements and capability constraints, but it is a highly researched topic. One current practical application of pre-combustion is the gasification of feedstocks, most commonly coal. Coal is oxidized under extreme pressure and temperature to produce synthetic fuel, which goes through the combustion process and produces less CO2 as a result.
Oxy-combustion
Oxy-combustion changes the combustion process by removing nitrogen, instead using pure oxygen to produce a cleaner result. Oxy-combustion is very much in the research and development phase, with few large-scale practical applications. The process calls for higher energy utilization to isolate oxygen, making it more expensive to put into practice. But there is promising research underway — most notably, technologies such as chemical looping combustion, which have the potential to completely remove CO2 from refinery emissions and use less energy in the process.
Read more: 5 key carbon capture technology trends for 2023 (with examples)
Methods of carbon utilization
Carbon utilization is in its early stages of development, and researchers are still exploring the most effective and efficient methods of use. Bioconversion and catalysis are currently the primary potential options.
Bioconversion
Bioconversion uses living organisms, such as microbes, to transform organic material into sources of energy. In the simplest sense, it’s planting trees, algae or bamboo to encourage natural photosynthesis for the reduction of CO2 in the atmosphere.
But artificial bioconversion is a slightly more complex process, and research is heavily invested in finding viable uses for the bioconversion of pollutants. One method targets carbon sequestration with carbon-fixing microbes that consume CO2 to build biomass, which is then converted into products like biofuel.
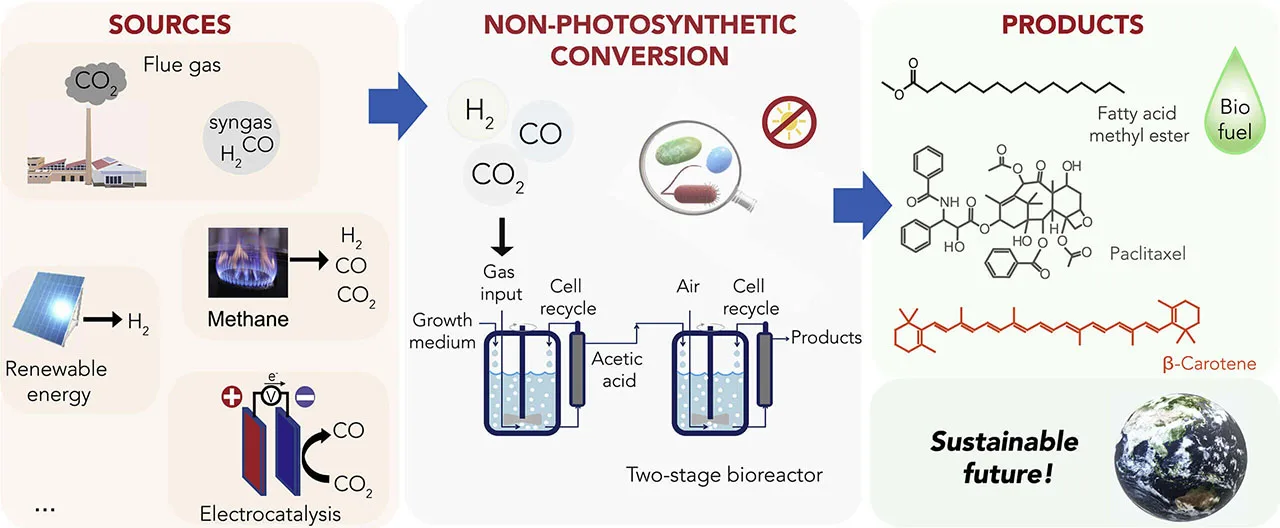
Source: "Light-Independent Biological Conversion of CO2" (Joule, Volume 4, Issue 10opens in new tab/window).
Practical applications of bioconversion in the energy industry are hard to come by because this method of carbon utilization is still undergoing R&D. It is currently most common within the pharmaceutical and food industries in the development and production of drugs and additives.
Catalysis
During catalysis, a catalyst is used to accelerate chemical reactions without getting damaged in the process, allowing it to be recycled. This has traditionally been performed via heterogeneous catalysis, where a solid catalyst on a porous support is used to convert fluid streams containing CO2.
More recently, research has investigated more complex catalytic systems which can utilize external energy sources to drive the reaction forward. These include photocatalysts, which utilize incoming photons to promote surface reactions, and catalysts for use in electrochemical cells. Electrolytic cells are seeing particular interest recently, with the reaction conditions at the anode and cathode influenced by the addition of catalysts and diffusion across the cell limited by the use of ionic exchange membranes.
Although research is ongoing, real wide-scale viable methods of splitting CO2 molecules through catalysis are still lacking because the procedure is often complex, expensive and energy-intensive. But breakthroughs, like a more efficient catalyst designed by Matteo Cargnello at Stanford Universityopens in new tab/window, are moving catalysis as a method of CO2 utilization forward. One currently viable application of catalysis occurs in the construction material industry, most notably in cement production.
Learn how Knovel's wealth of engineering information supports your energy transition.
The carbon storage process
Carbon storage in practical industry applications relies mostly on absorption, while adsorption remains in the research phase.
Absorption
Absorption involves capturing gaseous CO2 onto a liquid, most commonly a liquid amine scrubber. Liquid amine absorption is the current standard for the energy industry because it is available, well-researched and has been in practice for decades. It does have drawbacks, though; it is low-capacity, energy-intensive and inefficient within the capture process. From a sustainability perspective, absorption produces CO2 to capture and store CO2, making it a less-than-ideal long-term solution.
Adsorption
Adsorption refers to the storage of captured carbon within solid formations like zeolites, clay or nanocarbons. In the adsorption process, CO2 molecules are captured onto the solid support in a mechanism that is often defined by the surface area and availability of diffusion channels to access active surface sites. Adsorption can be performed physically, where the surface acts like a sponge and captures CO2 molecules, or chemically, where CO2 is converted into a chemical through a reaction. An example of chemical adsorption would be bonding calcium oxide with CO2 to create calcium carbonate.
Physical vs Chemical Adsorption
One of the defining differences between the two methods is longevity. Physical adsorption creates a weak hold, allowing CO2 to eventually return to its gaseous state, while chemical adsorption has the potential to hold onto CO2 indefinitely. Although adsorption is still in the R&D phase, it is seen as a better long-term solution for carbon storage because it is more efficient, allows for higher capacities and is easier to regenerate.
Determining a carbon storage site
Researching carbon storage sites comes with various considerations. Location is especially critical. Carbon storage is currently being done within five types of storage sites:
Saline formations
Depleted (and depleting) oil and natural gas reservoirs
Unmineable coal seams
Basalt formations
Organic shale formations
Saline formations and depleted or depleting natural gas reservoirs are the two most common carbon storage sites due to their availability and favorable conditions. The other types of storage sites are used on a smaller-scale basis.
Regardless of the type of site your organization chooses, a host of considerations should be made, including licensing requirements, leak and seismic activity potential and geologic properties like porosity and permeability. At the same time, governmental laws and regulations require equal attention.
For example, energy companies seeking to construct a storage site within the U.S need to obtain permits and authorization from the Environmental Protection Agency (EPA)opens in new tab/window throughout four key phases:
Pre-construction
Pre-injection
Injection
Post-injection
Each phase is a multi-step process that involves everything from site screening to applicant training and risk analysis. The process is designed to mitigate environmental risks and ensure the long-term efficacy of CO2 storage sites.
What is carbon accounting?
Carbon accounting is a facet of Life Cycle Analysisopens in new tab/window (LCA). It attempts to assess and account for the environmental impacts of manufactured goods through every stage of the process, from natural resources to finished products. Not only does carbon accounting measure the industry’s progress towards net-zero goals, but it also quantifies carbon reductions for the purpose of obtaining government grants, claiming tax credits and measuring return on investment.
Challenges of carbon accounting
In order to turn carbon waste into a commercially viable product, industries need a standardized method of accounting for carbon capture — a challenge in itself. While accounting for carbon poses numerous challenges, two commonly examined factors are the natural carbon cycle and formula standardization.
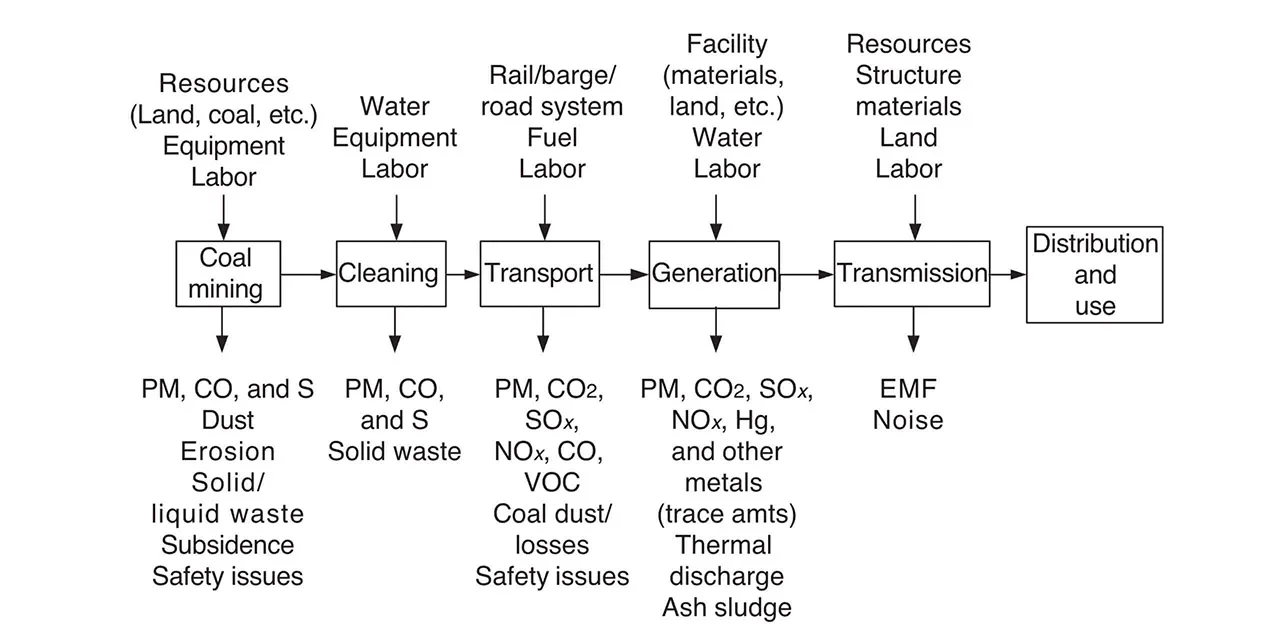
An LCA of electricity from coal from “Life Cycle Analysis of Power Generation Systems” (Encyclopedia of Energyopens in new tab/window).
The natural carbon cycle
One of the central challenges of carbon accounting is measuring exactly how much CO2 you’re actually removing from the atmosphere because of the natural carbon cycle. While organizations are employing technology to remove CO2, the environment is naturally employing its own system of complex processes.
For example, say a company tries to measure how much CO2 they are capturing in engineering ocean water. The ocean also has its own process for consuming and converting CO2 into metal carbonates like limestone — just one small fraction of the carbon cycle. These systems exist in equilibrium with each other, so removing carbon from the ocean impacts carbon concentrations in the air and on land. So, how do you measure what your CCUS technology captures during the process?
Multi-faceted formulas
Another key challenge lies within the standardization of a formula for carbon capture. Multiple questions arise, including:
Which factors go into the formula?
Do you account for the energy it takes to acquire resources?
Does the energy required for actually capturing, storing and utilizing CO2 go into the formula?
Should CO2 emissions from logistical aspects, like transportation of finished products, be included?
Do you measure how much CO2 you’re capturing or how much you’re removing? Or both?
All of these considerations, and countless more, make formulizing carbon accounting difficult. Adding to the challenge is the fact that the formulas must be globally — or at the very least, nationally — accepted in order to ensure organizations are following a standard set of procedures for CCUS.
Current solutions to the carbon accounting problem
Several organizations have attempted to create benchmarks for measuring captured carbon, with the most notable being the Intergovernmental Panel on Climate Change (IPCC).
The IPCC released the 2006 IPCC Guidelines for National Greenhouse Gas Inventoriesopens in new tab/window, which spans five volumes and includes detailed breakdowns of the entire CCUS process, as well as reporting methodologies. In 2019, the IPCC added a new methodology reportopens in new tab/window featuring updated research for use in conjunction with the 2006 guidelines:
Estimating, verifying & reporting emissions from CO2 storage sites
1. Site characterization | Confirm that geology of storage site has been evaluated and that local and regional hydrogeology and leakage pathways have been identified. |
2. Assessment of risk of leakage | Confirm that the potential for leakage has been evaluated through a combination of site characterization and realistic models that predict movement of CO2 over time and locations where emissions might occur. |
3. Monitoring | Ensure that an adequate monitoring plan is in place. The monitoring plan should identify potential leakage pathways, measure leakage and/or validate update models as appropriate. |
4. Reporting | Report CO2 injected and emissions from storage site. |
Source: Procedures for estimating emissions from CO2 storage sites from “2006 IPCC Guidelines for National Greenhouse Gas Inventories” Vol. 2 Chp. 5 (IPCCopens in new tab/window).
Government tax credits also require industries to follow certain guidelines. American-based organizations need to follow the instructions for Form 8933opens in new tab/window when applying for carbon tax credits, for example. Form 8933 also standardizes formulas for accounting for the capture, storage and utilization of carbon through industrial processes.
For example, Section 45Q(a)(1)opens in new tab/window allows “a credit of $20 per metric ton of qualified carbon oxide captured by you using carbon capture equipment that’s (1) originally placed in service at a qualified facility before February 9, 2018, (2) disposed of by you in secure geological storage, and (3) not used by you as a tertiary injectant in a qualified enhanced oil recovery (EOR) or natural gas recovery project or utilized by you in a manner described in section 45Q(f)(5).”
Even with the IPCC’s contributions to carbon accounting and governmental regulations for tax credits, industries still lack a global standardization of methodologies. At most, companies currently follow state or national guidelines or formulas devised by private carbon accounting firms. Standardizing the carbon accounting process is a key challenge for the future wide-scale deployment of CCUS technologies.
Incentives for utilizing CCUS technologies
Implementing CCUS technologies into your industrial processes can be complex, but a combination of short- and long-term benefits are available to incentivize organizations.
Enhanced oil recovery (EOR) is an incentive for energy companies right now because the technology and processes for EOR are well-researched and ready to implement. EOR is a technique used to extract oil from depleting reservoirs after primary and secondary techniques have been exhausted. But EOR has several drawbacks, most importantly, environmental impacts in the form of increased seismic activity. For this reason, carbon utilization is shifting toward the production of feedstocks rather than oil.
Organizations interested in implementing CCUS technologies into their workflow have two major short-term financial incentives to explore:
Carbon tax credits offset the cost of reducing carbon emissions through CCUS technologies and include financial incentives for installing carbon capture technologies, like Section 45Q(a)(1)opens in new tab/window in the U.S and CCUS tax credits additionsopens in new tab/window to the 2021 Canadian budget, among others globally.
Government grants aim to encourage energy companies to invest in CCUS research and practical projects through the Bipartisan Infrastructure Lawopens in new tab/window (U.S), Energy Innovation Programopens in new tab/window (Canada), Carbon Capture and Storage Infrastructure Fundopens in new tab/window (U.K) and many more around the world.
The goal of grants and tax credits is to incentivize companies to research ways to implement CCUS technologies in a cost-effective manner and help tip the economic balance in favor of carbon capture. But the long-term goal is to find ways to make carbon capture not only economically feasible but also profitable. The solution is CO2 utilization in the manufacturing of other products, like feedstocks and fuels, to turn carbon waste into a profitable commercial model.
Active CCUS projects and applications
Although carbon utilization isn’t (yet) a wide-scale operation, pioneering organizations across several industrial sectors have established successful businesses propped up by CCUS technologies.
Pioneering low-carbon energy
Iceland-based Carbon Recycling Internationalopens in new tab/window (CRI) has been producing “renewable methanol from carbon dioxide and hydrogen, for more sustainable fuels, chemicals and products” on an industrial scale since 2012. The company uses hydro and geothermal electricity to combine captured carbon with hydrogen and produce methanol, which is then sold for biodiesel or gasoline production. CRI invests in research and development of more efficient CCUS processes and works with global energy partners to put its product into practice.
Canadian Carbon Engineeringopens in new tab/window pioneered its Air to Fuels energy technology to produce carbon-neutral fuel with atmospheric captured carbon. Its process “integrates four growing fields – renewable electricity generation, Direct Air Capture, green hydrogen production, and sustainable fuel synthesis – to deliver a highly scalable, clean fuel solution. It delivers drop-in ready fuels that have a low lifecycle carbon intensity and are cost-competitive with biofuels.” The end result is a hydrocarbon fuel that’s immediately viable in practical applications and compatible with existing vehicle engines.
Building a carbon-negative world
Carbon8opens in new tab/window, based in the United Kingdom, has taken an entire CCUS facility and compressed it into two 40-foot containers that it calls the CO₂ntainers. According to Carbon8opens in new tab/window, “the CO₂ntainer captures CO₂ at the source, which becomes an ingredient to carbonate industrial residues destined for landfill. The solution is able to treat up to 12,000 tonnes of residues annually in the process.” The end product is a carbon-negative aggregate that has the potential for various practical applications, like cement production, roofing and road filler.
Canadian company CarbiCreteopens in new tab/window created a technology that allows concrete makers to manufacture “cement-free, carbon-negative concrete” by curing ground steel slag with captured carbon. According to CarbiCrete, the end product reduces cost by 10 to 20 percent and reduces 3 kilograms of CO2 per concrete masonry unit (CMU). The company makes implementation simple by managing the installation of the technology and the supply of materials for its partners.
Turning carbon waste into household products
Econicopens in new tab/window is a UK-based company that uses catalysis to convert CO2 into short-chain polymers with a unique technology that makes the processes less energy intensive. Not only is the process an effective utilization of CO2, but it's also economically efficient. According to Econicopens in new tab/window, the technology ensures that "up to 50% waste CO2 [is] used to replace fossil fuel-based feedstock," which means lower material costs and higher savings. The end product can be used to make everyday goods like insulation, adhesives and foams.
US-based Newlight Technologiesopens in new tab/window pioneered a groundbreaking technology based on the power of nature that uses “microorganisms found in the ocean to convert air and greenhouse gas (CO2e) into a natural biomaterial that we can melt and form into products: we call it AirCarbon.” The end product is a carbon-neutral, compostable, high-strength pellet that can be melted down to create a multitude of products ranging from furniture to electronics to packaging.
Follow us on LinkedIn for the latest engineering news.opens in new tab/window
Learn more about sustainable innovation with Elsevier
Read more insights into sustainable innovation and learn more about how Elsevier supports the world's leading engineering organizations.
October 2 webinar | Sustainability in Action: Green Chemistry for a Sustainable Future




